Reach out to us

- 415, rue Claude Nicolas Ledoux 13854
- AIX EN PROVENCE Cedex 3, France
- +33 (0)4 42 60 30 00
- contact@graftys.com

Graftys’ products are registered in more than 30 countries worldwide.

More than 80,000 units of injectable implanted since their launch.

More than 100 scientific papers about Graftys’s products and technologies have been publsihed on different prominent orthopedic journals and newspapers.
As an expert in Bone tissue engineering, we are committed to develop and commercialize solutions for stimulating and scaffolding bone regeneration after an injury. Graftys® technologies are intended for bony voids or defects that are not intrinsic to the stability of the bony structure. They are intended to be placed or injected into bony voids or gaps of the skeletal system (EU: orthopedic, cranio-facial, maxillo-facial, or periodontal; US: extremities and pelvis).

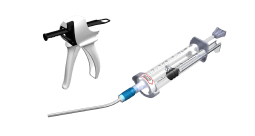
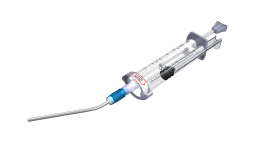
GRAFTYS® products are provided in a double-compartment mixing syringe, which is pre-filled with powder (composed of calcium-phosphate salts and a rheo-fluidifying polymer) and phosphate-based aqueous solution. The injection of GRAFTYS® products are performed manually or using the GRAFTYS® Delivering Gun. GRAFTYS® products are conditionned in a sterile, non-pyrogenic, single-use medical device available in 5cc, 8cc and 16cc (8cc*2), provided or not with a sterile cannula (Diam. 4 mm * Length 80 mm).
Eiffel Park 415, rue Claude Nicolas Ledoux Pôle d'Activités d'Aix en Provence 13854 AIX EN PROVENCE Cedex 3, France
Keep up-to-date with our latest news, events and congresses. Hope to see you soon at one of our events!